The evolving universe and its elements move and transfer energy from one end to another. It is no surprise that a metal rod heated at one end becomes completely heated. The perfume you sprayed can be smelled throughout the room, and the water, when heated from the bottom in a gyser, warms up the entire tank of water. This is no magic, it’s science; the kinetic molecular theory.
The kinetic molecular theory, also known as the kinetic particle theory, is one of the most important foundations in Physics and Chemistry. It explains how matter behaves at the microscopic level by describing how particles move, collide, and gain energy. From understanding why gas pressure increases in a heated cylinder to how perfume spreads across a room, the kinetic molecular theory connects science to everyday life in a surprisingly intuitive way.
This blog by VACE Global will help you clearly understand the complex concept of kinetic molecular theory, its real-life applications, and its behaviour with the three states of matter. This concept is a part of chemistry in every board, whether Edexcel or Cambridge.
For more well-explained O-level, A-level, and IGCSE revision notes, stay connected with VACE Global.
Key Takeaways:
- The kinetic molecular theory explains how particles in solids, liquids, and gases move and gain energy.
- Temperature increases particle kinetic energy, affecting observable properties like pressure, expansion, and diffusion.
- Particle arrangement and spacing help differentiate solids (vibrate), liquids (flow), and gases (spread out rapidly).
- KMT underpins major gas laws and thermodynamics, linking microscopic particle motion to measurable quantities.
- Although idealized, KMT has real-world applications in daily life and industry, from perfume diffusion to refrigeration and tyre pressure.
What is the Kinetic Molecular theory?
The Kinetic Molecular Theory (KMT) explains the states of matter. It defines how every matter is composed of tiny particles. These particles are always in motion.
The KMT explains the observable behaviour of particles that was experimentally discovered. It is more often referenced in the behavior of gases in any experiment, but it is also applied to solids and liquids.
The kinetic molecular theory states that the motion of a molecule in any state of matter, either solid, liquid, or gas, is predicted on the variation of factors such as temperature, volume, and pressure of the atmosphere.
The origin:
The earliest ideas behind the kinetic molecular theory can be traced back to Daniel Bernoulli in 1738. In his work Hydrodynamica, he suggested that gases are made up of tiny particles in constant motion, and that gas pressure results from their collisions with container walls. He also linked temperature to the kinetic energy of these particles — a concept that was far ahead of its time and not immediately accepted.
Over the late 18th and early 19th centuries, scientists such as Mikhail Lomonosov, Georges-Louis Le Sage, John Herapath, and John James Waterston expanded on Bernoulli’s ideas. The theory took a major step forward in 1857 when Rudolf Clausius introduced the concepts of rotational and vibrational motion in gas molecules and explained the mean free path. His refinements helped connect microscopic particle motion to the observable properties of gases, forming the basis of the modern kinetic molecular theory.
Objectives of kinetic molecular theory:
The kinetic molecular theory (KMT) helps explain how matter behaves by describing it in terms of tiny particles (atoms or molecules) that are always in random motion. According to this theory, the average kinetic energy of particles depends on temperature, which allows scientists to link microscopic motion to macroscopic properties such as pressure, temperature, and volume. This makes the theory especially useful for understanding gases and the differences between solids, liquids, and gases.
Key Purposes of KMT
- Explaining observable properties:
The theory connects what we can measure — like pressure or temperature — to particle movement and collisions at the microscopic level. - Modelling gas behaviour:
Gases are described as particles that are far apart, have negligible volume, move randomly, and collide elastically. This explains why gases fill their containers, why pressure changes with temperature, and why temperature reflects the average kinetic energy of particles. - Differentiating states of matter:
KMT helps distinguish solids, liquids, and gases by describing differences in particle arrangement and motion:
- Solids: tightly packed particles that vibrate in fixed positions
- Liquids: particles that move more freely and slide over one another
- Gases: particles that are widely spaced and move rapidly in all directions
- Solids: tightly packed particles that vibrate in fixed positions
- Predicting particle motion:
The theory also explains phenomena such as diffusion (spreading of smells or gases) and effusion (gas escaping through small openings), which depend on particle speed. - Forming a scientific framework:
KMT provides the conceptual basis for understanding the Ideal Gas Law (PV = nRT) and how gases respond to changes in temperature, volume, and pressure.
Kinetic theory of matter: The foundation
To understand the kinetic molecular theory of gases, it’s important to begin with its broader idea, the kinetic theory of matter. This theory provides a scientific model that helps us visualize and explain how matter behaves at a microscopic level, even though we cannot directly observe particles with the naked eye.
The kinetic theory of matter states that:
All matter is made up of tiny particles (atoms or molecules) that are always in motion.
This movement exists in every substance, regardless of whether it is a solid, liquid, or gas. However, the way particles move varies depending on the state of matter, because each state has different amounts of energy and different levels of particle spacing.
How Particles Behave in Different States of Matter
The theory explains the three classical states of matter as follows:
- Solids:
In solids, particles are packed very closely together. They cannot move freely, so they vibrate in fixed positions. This close arrangement is why solids have a definite shape and volume, and why they are not easily compressed. - Liquids:
Particles in liquids have more space and energy than in solids. While still close together, they can slide past one another, which allows liquids to change shape and take the form of their container. However, they still maintain a definite volume. - Gases:
Gas particles have the most energy and are far apart, moving freely and rapidly in all directions. Since there is a lot of space between particles, gases can expand, fill the entire container, and be easily compressed.
Temperature and Particle Energy
A key idea behind the kinetic theory of matter is that the energy of particles increases as temperature increases. Temperature acts as a measure of average kinetic energy — so heating a substance gives its particles more energy to move.
This explains everyday observations such as:
- Heated solids soften because increased vibration weakens their structure.
- Liquids evaporate or boil because fast-moving particles break away from the surface.
- Gases expand because particles move faster and spread out more.
These examples help students connect the theory to real-world phenomena, which is particularly valuable for exam understanding in Cambridge O Level and A Level Physics and Chemistry.
Postulates of Kinetic Molecular Theory:
The kinetic theory of gases explains how gases behave by describing their particles and motion at a microscopic level. According to this theory, gases are made up of extremely small particles that are far apart, move randomly in straight lines, and collide with each other and with the walls of their container. Their collisions are elastic, meaning no kinetic energy is lost, and the average kinetic energy of gas particles depends directly on the absolute temperature. These ideas help us understand why gases can be compressed, why they exert pressure, and how temperature affects gas behaviour.
Key Postulates of the Kinetic Theory:
- Particle Nature & Spacing: Gas molecules are tiny compared to the space between them, so their actual volume is negligible relative to the volume occupied by the gas.
- Constant Random Motion: Gas particles move continuously, rapidly, and randomly in all directions. They travel in straight lines until they collide with another particle or with container walls.
- Negligible Intermolecular Forces: Under normal conditions, gas particles do not attract or repel one another significantly, except during collisions.
- Elastic Collisions: Collisions between gas particles and between particles and container walls are perfectly elastic, meaning no kinetic energy is lost through the collision.
- Kinetic Energy & Temperature: The average kinetic energy of gas particles is directly proportional to the absolute temperature (measured in Kelvin). As the temperature increases, particles move faster.
Implications of the Theory
- Compressibility: Because there is so much space between gas particles, gases can be compressed much more easily than solids or liquids.
- Pressure Formation: Gas pressure results from particles hitting the walls of their container. More frequent or more forceful collisions increase pressure.
- Effect of Temperature: Heating a gas increases particle speed and kinetic energy, which can increase pressure or cause the gas to expand.
How does KTM explain states of matter?
Although the kinetic molecular theory (KMT) is often discussed in the context of gases, it also provides a useful framework for understanding how particles behave in solids, liquids, and gases. By focusing on particle arrangement, spacing, and kinetic energy, KMT helps explain why different states of matter have different physical properties.
According to KMT:
The state of matter depends on how closely particles are packed together and how much kinetic energy they possess.
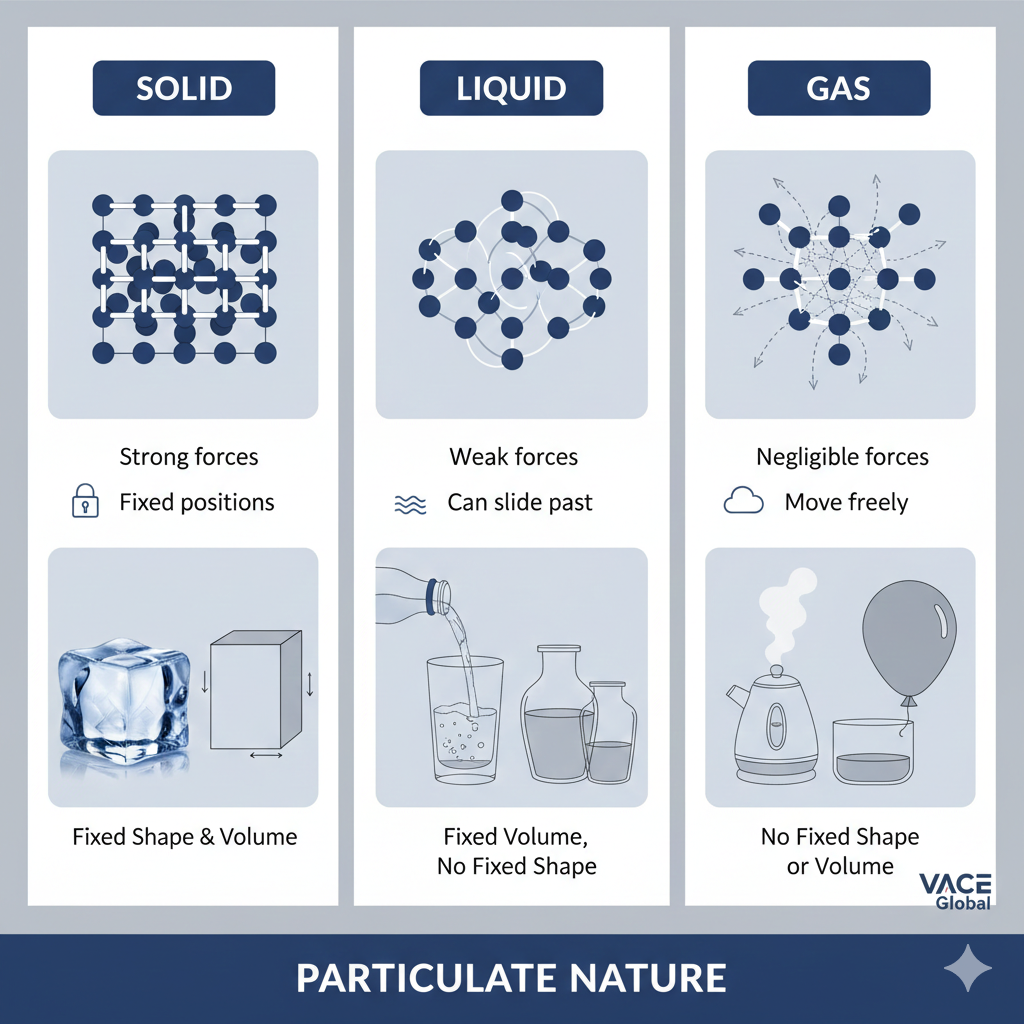
Solids:
In solids, particles are packed very closely together in a fixed, orderly pattern. They do not have the freedom to move around but can vibrate about fixed positions. This limited motion is due to strong intermolecular forces and low kinetic energy.
Because of this:
- Solids have a definite shape
- Solids have a definite volume
- They are not affected by the shape of their container
The structured arrangement explains why solids are rigid and why they cannot be compressed easily.
Liquids:
Particles in liquids have more kinetic energy compared to particles in solids. They are still fairly close together but can slide or flow past one another, allowing liquids to move and adapt to the shape of a container.
As a result:
- liquids take the shape of their container
- liquids have a fixed volume
- They are slightly more compressible than solids (but still resist compression)
This intermediate behaviour reflects a balance between particle mobility and intermolecular attraction.
Gases:
Gas particles have the highest kinetic energy among the three classical states. They are far apart, move rapidly, and travel in straight lines until they collide.
At this level of motion:
- Gases take both the shape and volume of their container
- Gases are highly compressible
- They spread out (diffuse) quickly because particles can overcome intermolecular forces
This explains familiar gas behaviours such as perfume spreading across a room or air filling a balloon.
Real-World Applications of Kinetic Molecular Theory:

Although KMT is theoretical, its applications are visible in everyday life. Examples include:
- Perfume spreading in a room (diffusion)
- Tyre pressure increases on hot days
- Refrigeration and air-conditioning systems
- Balloon expansion, contraction, and leakage
- Aerosol sprays and inhalers
- Breathing mechanisms (respiration)
- Vapour pressure and boiling
On a larger scale, KMT underpins fields like chemical engineering, meteorology, physical chemistry, and thermodynamics.
Limitations of the Kinetic Theory:
The kinetic molecular theory assumes ideal behaviour, which is not always realistic. Limitations include:
- Real gases experience intermolecular forces, especially at high pressure and low temperature.
- Gas particles occupy actual volume, which becomes significant under extreme conditions.
- Elastic collisions are an approximation.
- Deviations lead to the need for real gas models, such as the van der Waals equation.
Understanding these limitations allows students to distinguish between ideal and real gas behaviour, an important idea in higher-level physics and chemistry.
Conclusion:
The kinetic molecular theory provides a powerful model for explaining how particles behave in solids, liquids, and gases. From basic concepts like diffusion and pressure to advanced applications like gas laws and thermodynamics, KMT connects microscopic particle motion to everyday phenomena and scientific principles. For students, mastering this theory strengthens conceptual understanding in both physics and chemistry and offers clearer insight into how matter behaves in the real world.
How does VACE Global help in understanding the kinetic theory?
At VACE Global, we prioritize conceptual clarity, a key requirement for mastering scientific topics like the kinetic molecular theory. With support from our expert tutors, students learn through real-world examples, visual explanations, and exam-focused instruction aligned with the Cambridge O Level and A Level syllabus. To strengthen exam preparation further, students can also practice using past papers, helping them apply concepts and develop problem-solving confidence. Our personalized online learning environment is designed to support academic improvement and help students perform at their best.
If you want to build a stronger foundation in Physics and Chemistry and excel in exams, now is the perfect time to take action. Enroll now and start learning with top-tier tutors who know how to make complex scientific ideas simple, understandable, and exam-ready.
Glossary:
- Kinetic Energy: energy of motion
- Ideal Gas: a gas that obeys kinetic assumptions perfectly
- Pressure: force per unit area due to particle collisions
- Diffusion: spreading of particles
- Temperature: measure of average kinetic energy
FAQs:
Q1: What is the kinetic molecular theory in simple words?
It explains how particles of matter move, gain energy, and interact.
Q2: What are the postulates of kinetic molecular theory?
Tiny particles, random motion, elastic collisions, no intermolecular forces, and energy depend on temperature.
Q3: Why do particles move faster when heated?
Because temperature increases kinetic energy.
Q4: How does kinetic theory relate to gas laws?
It provides molecular explanations for Boyle’s, Charles’, and other gas laws.
Q5: Is the kinetic molecular theory part of the Cambridge syllabus?
Yes, it appears in multiple CAIE O Level and A Level Physics & Chemistry topics under states of matter, matter & thermal physics, and gas laws.
